What Is the Fundamental Difference Between Covalent and Ionic Bonds
In an ionic bond one partner accepts electrons from the other. Ionic and covalent bonding involve electrons.

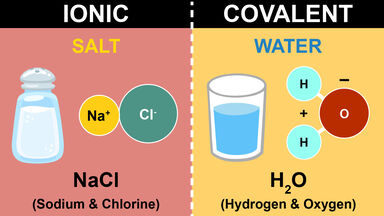
Ionic Bond Vs Covalent Bond Venn Diagram Shows The Similarities And Differences Between The Chemical Bonds Click Covalent Bonding Ionic Bonding Chemical Bond
In a covalent bond the partner share a pair of electrons.
. In an ionic bond one partner accepts electrons from the other. 17 what is the fundamental difference between covalent and ionic bonding a covalent bonding involves only the outermost electren shell ionic bonding alo b covalent bonds form between atoms of the same element. The difference between covalent and ionic bonding is that covalent bonding is a type of chemical bond that involves the sharing of pairs of electrons between atoms.
13What is the fundamental difference between covalent and ionic bonding. In an ionic bond one partner accepts electrons from the other. In an ionic bond one partner excepts electrons from the other.
In ionic bonding one partner does and the other does not. Ionic bonds do not use the octet rule. However by doing this both bonds create elements that are neutrally charged making them stable compounds.
A covalent bond is sharing of two electrons between two atoms something mutual. While there exists different types of chemical bonds our focus in this article is to discuss the difference between Ionic and Covalent bonds. In a covalent bond the partners share a pair of electrons.
In covalent bonding both partners end up with filled outer electron shells. In a covalent bond the partners share a pair of electrons. In an ionic bond one of them is more electronegative.
Methane molecule CH4 also offers covalent bonds between carbon and hydrogen atoms you will find four covalent bonds between one central carbon atom and 4 hydrogen atoms four C-H bonds. One loses its electron to the other. Ionic bonds occur between two species which are electrostatically attracted towards each other whereas covalent atoms bond covalently through the sharing of electrons between their outer shells.
While in an ionic bonding there is only one electron involved between two highly electronegative and electropositive atoms. Ionic bonds form between atoms of c in a covalent bond the partners share a pair of electrons. The key difference between covalent and noncovalent bonds is that covalent bonds form when two atoms share their electrons with each other whereas noncovalent bonds form either by completely exchanging electrons between two atoms or by not exchanging any electron.
In ionic bonding one partner does and the other does not. The main difference between covalent and ionic bonds is that ionic bonds occur between two species which are electrostatically attracted towards each other whereas covalent bonds occur covalently through the sharing of electrons between their outer shells. In an ionic bond one partner accepts electrons from the other.
A In a covalent bond the partners share a pair of electrons. B In covalent bonding both partners end up with filled outer electron shells. The covalent bond and the ionic bond.
What are the rules for determining if a. Expansion of the ideal covalent and ionic models leads chemists to the concepts of electronegativity and polarizability and thus to the classification of polar and non-polar bonds. One shares and the other trades.
Ionic bonding involves sharing of protons. What is the fundamental difference between covalent and ionic bonding. In a covalent bond the partners share a pair of electrons.
Involves the loss and gain of electrons form ions. Key Differences Between Covalent Metallic and Ionic Bonds. So while they are different ionic and covalent bonds have a few.
Ionic bonds form between atoms of different elements. Covalent bonding involves only the outermost electron shell. An ionic bond essentially donates an electron to the other atom participating in the bond while electrons in a covalent bond are shared equally between the atoms.
An ionic bond essentially donates an electron to the other atom participating in the bond while electrons in a covalent bond are shared equally between the atoms. C In covalent bonding both partners end up with. Ionic bonds form between a metal and a nonmetal.
Group of answer choices. Additionally both bonds focus on the electrons. A In a covalent bond the partners share a pair of electrons.
There are four major types of chemical bonds. Covalent bonds ionic bonds hydrogen. What is the fundamental difference between covalent and ionic bonding.
Ionic bonding also involves the next electron shell inside the. Ionic bonding also involves the next electron shell inside the. Covalent bonding involves sharing of electrons.
In ionic bonding one partner does and the other does not. C Covalent bonding involves only the outermost electron shell. Ionic bonding on the other hand is a type of chemical bond in which one atom donates one or more electrons to another atom.
Ionic and covalent bonds are fundamentally different in the way they are formed. A In a covalent bond the partners have identical electronegativity. The Ionic bonds tend to be formed between a metal and a non.
Covalent bonds form between atoms of the same element. Covalent bonds form based on the octet rule. Up to 256 cash back What is the fundamental difference between covalent and ionic bonding.
This is the main difference between Ionic and Covalent Compounds. B In covalent bonding both partners end up with filled outer electron shells. In a covalent bond the partners share a pair of electrons.
The key difference between ionic and covalent bonds is that ionic bonds occur between atoms having very different electronegativities whereas covalent bond. In an ionic bond one partner captures an electron from the other. Covalent bonding involves the sharing of electrons.
In an ionic bond one partner accepts. 7 rows An ionic bond essentially donates an electron to the other atom participating in the bond. The concept of chemical bonding is normally presented and simplified through two models.
B In a covalent bond the partners share a pair of electrons. In an ionic bond one partner accepts electrons from the other. This new canonical model which is a force-based approach with a basis in fundamental molecular quantum mechanics confirms much earlier assertions that in fact there are no fundamental distinctions among covalent bonds ionic bonds and intermolecular interactions including the hydrogen bond the halogen bond and van der Waals interactions.
In an ionic bond one partner accepts electrons from the other. Covalent bonds form between two nonmetals. In general metallic elements tend to form ionic bonds and non-metallic elements tend to form covalent.
The only pure covalent bonds occur between identical atoms.
What S The Difference Between An Ionic Bond And A Covalent Bond Quora

Covalent Bonding Google Search Chemistry Education Teaching Chemistry Chemistry Basics
Comments
Post a Comment